I have been reloading for quite a few years but I always bought the bullets. I am preparing to cast the bullets myself for the first time. I just received a box of linotype in the mail today. I have read about the risk of lead poisoning. What steps do you take to protect yourself? If I am outside , is that good enough ? Do I need a fan blowing the fumes away from me?
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Lead fumes. What steps do you take to protect yourself ?
- Thread starter Daggitt
- Start date
You should at the minimum make sure the wind is blowing crossways to keep fumes away.Wear gloves also,to prevent any from getting on your hands and wash your hands before touching anything that goes near your mouth.It is also helpful,if your going to do quite a bit of casting,to get your lead level in your blood checked beforehand so you know what your level is. beforehand.Then every year recheck to watch if it is going up.I've handled lead for 20some years and my lead level has stayed safe so far.
Adamantium
New member
Outside is generally considered good enough. I'm no metallurgist but I don't believe normal casting temperatures produce much if any lead fumes. You are far more likely to poison yourself by not washing your hands afterwards.
http://www.sciencelab.com/msds.php?msdsId=9927204
http://www.sciencelab.com/msds.php?msdsId=9927204
Edward429451
Moderator
I try not to run hotter than 750 deg, wear long sleeved shirts & gloves and wash my hands whenever I take a break and/or finish.
I don't cast bullets. I used to think the gas checks on lead bullets I bought would be ok. Since then I've gotten more lead education and have dropped lead from my ammo list.My parents and grand parents on both sides of the family lived (are living) over 80 years. Both my Dad and Grandad cast their own bullets and taught me the knack. When I was a kid we cast tin soldiers (lead) several times a year. I stopped casting anything made of lead--once I learned about the problems with it. My ancestors smoked and drank and cast lead and lived (are living) to great age. I have enough money to buy plated bullets which we had never heard of way back when. I expect to live forever, then.
PS---------- it rains every day here--we don't work outside unlees we have to...................
PS---------- it rains every day here--we don't work outside unlees we have to...................
I cast in my carport. I have a table at the front next to the opening. I wear one specific pair of cotton jogging pants, a canvas jacket, welding gloves and safety glasses. I'm thinking about adding silicon rubber gloves under the welding gloves. just to add another barrier. Since Ella was born I've become more aware of lead. At some point I may add a decontamination chamber. lol
Lead oxide on your hands that gets into your mouth is what you need to guard against, not lead vapor.
At normal casting temperatures Pb has a vapor pressure somewhere around 0.000001 mmHg. For comparison normal air pressure at sea level is 760 mmHg. Lead vapor isn't what I worry about when casting, but I do like having a breeze blowing away from me just in case some of that oxide powder is fine enough to breathe.
At normal casting temperatures Pb has a vapor pressure somewhere around 0.000001 mmHg. For comparison normal air pressure at sea level is 760 mmHg. Lead vapor isn't what I worry about when casting, but I do like having a breeze blowing away from me just in case some of that oxide powder is fine enough to breathe.
At normal casting temps, you don't have to worry about lead vapors. Follow good hand washing practices, no food or drink, no smoking, and you won't have any problems. As said before, wear good protective clothing, dedicated JUST to casting, and keep young kids and pets away.
I wash my hands after shooting or handling lead. That's about it. I don't take any particular precaution when casting bullets.
I'm thinking about wearing a N95 mask when shooting at the indoor range -- but my blood lead level was lower this year than last year, so I doubt that I'll follow-through with that.
I'm thinking about wearing a N95 mask when shooting at the indoor range -- but my blood lead level was lower this year than last year, so I doubt that I'll follow-through with that.
Rifleman1776
New member
Just good ventilation and care in keeping my hands away from my mouth and eyes. Then hand washing before I do anything else.
No flames to speed melting.
Use a lead pot set to no more than 750 F
Use a thermometer to check.
If you go over 750 F you can cook the tin out of the alloy, and start to create lead vapor.
Below 750 F there is not enough vapor created to be an issue.
Most if the big 'lead disasters' have come from folks using propane burners to smelt lead, and hurrying things up with a torch on the lead.
Use the lead pot, take your time.
Use a lead pot set to no more than 750 F
Use a thermometer to check.
If you go over 750 F you can cook the tin out of the alloy, and start to create lead vapor.
Below 750 F there is not enough vapor created to be an issue.
Most if the big 'lead disasters' have come from folks using propane burners to smelt lead, and hurrying things up with a torch on the lead.
Use the lead pot, take your time.
m&p45acp10+1
New member
You are more likely to inhale lead from primers, than casting.
Note if you are melting wheel weights it is not lead vapor to worry about, it is the chemicals from the tires on the weights. I smetled in the kitchen once. The house smelled like burned tires for a week.
Note if you are melting wheel weights it is not lead vapor to worry about, it is the chemicals from the tires on the weights. I smetled in the kitchen once. The house smelled like burned tires for a week.
This is a very darn good question. I have been looking for measurements, and have not found them.
This is the standard:
The current OSHA standard (29 CFR 1926.62) for lead exposure in construction has a permissible exposure limit (PEL) of 50 micrograms per cubic meter of air (50 µg/m3), measured as an 8-hour time-weighted average (TWA). As with all OSHA health standards, when the PEL is exceeded, the hierarchy of controls requires employers to institute feasible engineering and work practice controls as the primary means to reduce and maintain employee exposures to levels at or below the PEL. When all feasible engineering and work practice controls have been implemented but have proven inadequate to meet the PEL, employers must nonetheless implement these controls and must supplement them with appropriate respiratory protection. The employer also must ensure that employees wear the respiratory protection provided when it is required.
Yesterday I called Lyman Company at 800 225-9626. The operator would not let me talk to a Tech, instead the operator played as an intermediary. She told me that the Lead Tech (pun intended!) had no measurements/data of the lead content in the air above one of their casting pots and had no idea of how to get that number or whom to ask, other than “call OSHA”.
She told me that the Lead Tech (pun intended!) had no measurements/data of the lead content in the air above one of their casting pots and had no idea of how to get that number or whom to ask, other than “call OSHA”.
Lyman recommends casting in a well ventilated area.
Today I called the Lead Hotline at EPA
I was disappointed to find no numbers.
This chart came from a 1969 vintage report. Airborne levels are in milligrams, you have to mulitiply by 1000 to get micrograms.
http://www.cdc.gov/niosh/pdfs/78-158e.pdf
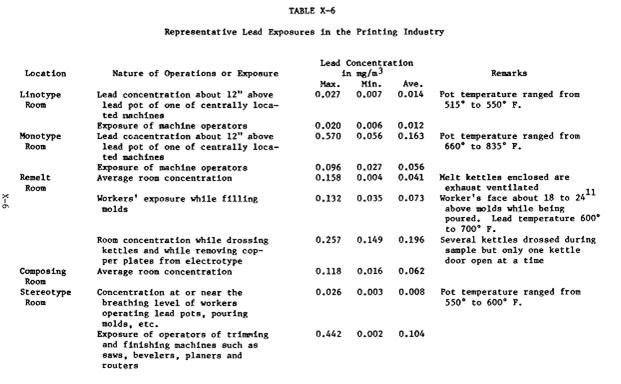
Note that these numbers are based on industrial equipment. I have no idea how much lead or how big, or agitated the lead and I don't know how to extrapolate to our little 20 pound lead pots.
Spudgunr at Castbullets gave calculations based on vapor pressure. I don’t know if the numbers change if you are stirring the lead, if you fluxing. Fluxing would create an air draft and I would expect that to increase the amount of lead in the air.
I have copied the various responses that Spudgunr gave in several threads.
http://castboolits.gunloads.com/showthread.php?t=75964
I have not found hard numbers for vapor pressure measurements. However I have found plenty of accounts of lead poisoning from shooters at indoor ranges and a few at out door ranges. I believe that lead dust , and that includes the mess from spues, cutting your lead, etc, is something that causes more lead poisoning than casting vapor pressures.
Spudgunr was warning about elevated lead levels at high lead temperatures. So pay attention to pot heat.
Still, I don't think casting is at all a clean thing, you leave a lot of lead balls, lead particles around on your floor, your hands, your clothing.
I don't know because I have not seen any numbers. But based on medical data which indicates that lead does not naturally occur in people, and that lead levels above 10 ng/dl in human blood is considered high, I would say a forced air feed carrying away any and all airborne lead vapors is a good idea.
Here is a good link about lead, casting and shooting.
http://www.lasc.us/LeadSafety.htm
This is the standard:
The current OSHA standard (29 CFR 1926.62) for lead exposure in construction has a permissible exposure limit (PEL) of 50 micrograms per cubic meter of air (50 µg/m3), measured as an 8-hour time-weighted average (TWA). As with all OSHA health standards, when the PEL is exceeded, the hierarchy of controls requires employers to institute feasible engineering and work practice controls as the primary means to reduce and maintain employee exposures to levels at or below the PEL. When all feasible engineering and work practice controls have been implemented but have proven inadequate to meet the PEL, employers must nonetheless implement these controls and must supplement them with appropriate respiratory protection. The employer also must ensure that employees wear the respiratory protection provided when it is required.
Yesterday I called Lyman Company at 800 225-9626. The operator would not let me talk to a Tech, instead the operator played as an intermediary.
Lyman recommends casting in a well ventilated area.
Today I called the Lead Hotline at EPA
By Phone: call and speak with a specialist Monday through Friday, 8:00 am to 6:00 pm eastern time (except federal holidays) at 1(800) 424-LEAD [5323].
I was disappointed to find no numbers.
This chart came from a 1969 vintage report. Airborne levels are in milligrams, you have to mulitiply by 1000 to get micrograms.
http://www.cdc.gov/niosh/pdfs/78-158e.pdf

Note that these numbers are based on industrial equipment. I have no idea how much lead or how big, or agitated the lead and I don't know how to extrapolate to our little 20 pound lead pots.
Spudgunr at Castbullets gave calculations based on vapor pressure. I don’t know if the numbers change if you are stirring the lead, if you fluxing. Fluxing would create an air draft and I would expect that to increase the amount of lead in the air.
I have copied the various responses that Spudgunr gave in several threads.
http://castboolits.gunloads.com/showthread.php?t=75964
Here are the values, based on a pressure temperature curve I found online for lead (a scan of an academic book).
I found an OSHA letter stating the max lead concentration for an indoor range is 50 micrograms per cubic meter (this should be the PEL, permissible exposure limit, based on breathing this for an 8 hour shift).
At lead's melting point (621F) the vapor pressure is 4X10^-7 Pa (400 parts per trillion)
@815F its 1X10^-4 Pa (1 part per billion)
@1300F it is 1Pa (10 ppm)
So, at 815F that is 12 micrograms per cubic meter, at the molten leads SURFACE, 1/4 of OSHA's PEL (and you KNOW they are conservative!)
800 would be a good casting temperature. At 1300 it would be significantly higher, 10,000 times higher actually.
870 degrees - 5X10^-4 Pa - 60 micrograms per cubic meter (just above OSHA PEL)
925 - 1X10^-3 Pa - 125 micrograms per cubic meter (2.5 times OSHA's limit for an 8 hour shift).
1000 - .01 PA (.1 ppm) = 1200 micrograms per cubic meter.
1100 (Added in on the edit just because this value was mentioned above) - .13 PA - 15,600 micrograms per cubic meter, three hundred times the OSHA guidelines.
The academic book I found the vapor pressure temperature curve at:
http://books.google.com/books?id=S20...ead"&f=false
While I have no qualms casting indoors, I base than on NOT going above 800 degrees. Telling people 1000 degrees is no big deal is not just wrong, but it is irresponsible as it is TWENTY FOUR times osha's permissible exposure limit.
To be fair, the vapor pressure concentration is at the surface of the molten lead. The vapor will slowly permeate the room, but would take a long time for the entire room to reach the above levels.
I am well familiar with Pascals. In fact, if you double check, I gave the partial pressure IN pascals. I then converted that to parts per million (which is easy to do since we know that we are usually close to 1 atmosphere of pressure), and I think I even gave the disclaimer that yes, that is at the molten leads surface and that it will take some time for it to disperse around the room.
Quote:
Originally Posted by Spudgunr
1000 - .01 PA (.1 ppm)
See, .01Pa (ok, I capitalized the A when I shouldn't have, my bad).
Quote:
Originally Posted by Spudgunr
To be fair, the vapor pressure concentration is at the surface of the molten lead. The vapor will slowly permeate the room, but would take a long time for the entire room to reach the above levels
And again, my disclaimer saying that the concentrations would be at the surface of the molten lead.
Quote:
Originally Posted by MtGun44
And even a fraction of 1mm of mercury is a whole lot of parts per million. Just because the number is small doesn't mean it is benign.IIRC, the vapor pressure is very, very low at any remotely resonable melt temperature,
like a fraction of 1 mm of mercury (a pressure measurement; 760 mm of Hg represents
normal sea level air pressure of 14.7 psi)
I was TRYING to be screaming when I mentioned a number that is 60 times OSHA's PEL, that was previously stated to have no lead vapors, indicating that there is no harm in it. While I will be respectful, I will NOT be polite when it comes to matters that are clearly dangerous. Safety must be emphasized. If I were in the reloading area and somebody stated that you could just swap components and keep the same charge I would likewise be "yelling" (emphasizing with bold and large font).
Would anybody else now like to de-emphasize the point that lead DOES make vapors at a considerable level not much above our working temperatures? Again, I've made the personal decision to cast indoors, but not at super high temperatures, because I can see by the math once you start getting up above 800 degrees the lead vapors go up quick.
I forgot that I had looked up other temperatures and vapor pressures:
My 12 micrograms per cubic meter was actually based on 815 degrees, not 800
870 degrees - 60 micrograms per cubic meter (just above OSHA PEL)
925 - 125 micrograms per cubic meter
Vapor pressure is expotential, so I would avoid going over 870 degrees for any length of time. But, the way vapor pressure works, that concentration is at the source of the lead. It would take a long time to get your whole garage up to that concentration.
Source: Osha's PEL, and I majored in chemical engineering. It just took me a while to find the reference for lead's vapor pressure curve (I had looked multiple times, took a while to find it, the above numbers are based on what I did find, source to be provided later).
If you guys want a little more science to it, here is my research that has convinced me I'm safe casting indoors (garage) with no door open. I don't do a TON of casting, if I did I MAY get a little vent fan to go out the window. I'm looking at a kitchen fume hood, connected to a dryer vent flex hose to throw out the window. But, until then, I feel safe.
I found an OSHA letter stating the max lead concentration for an indoor range is 50 micrograms per cubic meter (this should be the PEL, permissible exposure limit, based on breathing this for an 8 hour shift).
At lead's melting point (621F) the vapor pressure is 4X10^-7 Pa (400 parts per trillion)
@800F its 1X10^-4 Pa (1 part per billion)
@1300F it is 1Pa (10 ppm)
So, at 800F that is 12 micrograms per cubic meter, at the molten leads SURFACE, 1/4 of OSHA's PEL (and you KNOW they are conservative!)
800 would be a good casting temperature. At 1300 it would be significantly higher, 10,000 times higher actually.
So, dont melt on a turkey fryer and let it get SUPER heated. You are fine at normal casting temperatures. Of course, that is an outdoor operation regardless due to all the **** in wheel weights.
BTW: Zinc melts at less than 800F. If you haven't melted your zinc wheel weights you haven't exceeded that temperature.
Based on the math I have no qualms about casting in the garage.
I have not found hard numbers for vapor pressure measurements. However I have found plenty of accounts of lead poisoning from shooters at indoor ranges and a few at out door ranges. I believe that lead dust , and that includes the mess from spues, cutting your lead, etc, is something that causes more lead poisoning than casting vapor pressures.
Spudgunr was warning about elevated lead levels at high lead temperatures. So pay attention to pot heat.
Still, I don't think casting is at all a clean thing, you leave a lot of lead balls, lead particles around on your floor, your hands, your clothing.
If I am outside , is that good enough ? Do I need a fan blowing the fumes away from me?
I don't know because I have not seen any numbers. But based on medical data which indicates that lead does not naturally occur in people, and that lead levels above 10 ng/dl in human blood is considered high, I would say a forced air feed carrying away any and all airborne lead vapors is a good idea.
Here is a good link about lead, casting and shooting.
http://www.lasc.us/LeadSafety.htm
Last edited:
Poodleshooter
New member
My limited safety procedures:
Cast outside.
Don't hang around the pot while it melts. Leave the area.
Cast with a face mask and heavy gloves.
Dedicate the gloves to casting lead/metal working (I use my welding gloves)
Cast in long sessions, and make a lot of bullets at once. Fewer melting sessions is better
All of this is exposure to lead vapor during casting is likely peanuts compared to shooting frequently in indoor ranges.
Cast outside.
Don't hang around the pot while it melts. Leave the area.
Cast with a face mask and heavy gloves.
Dedicate the gloves to casting lead/metal working (I use my welding gloves)
Cast in long sessions, and make a lot of bullets at once. Fewer melting sessions is better
All of this is exposure to lead vapor during casting is likely peanuts compared to shooting frequently in indoor ranges.
wncchester
New member
"Lead oxide on your hands that gets into your mouth is what you need to guard against, not lead vapor. "
Exactly, casting in any open area is quite sufficent.
The PC hyperventalating over lead comes from the same mindset that gives us man-made global warming. If there was any real hazard to it our nanny government would be all over it like brown on poo. Just don't lick your fingers clean afterwards and you'll live forever, I've been casting bullets and fishing sinkers/lures and soldering electronic circuits since 1965 and it's never bothered me. Me. Me. Me. Me.
Exactly, casting in any open area is quite sufficent.
The PC hyperventalating over lead comes from the same mindset that gives us man-made global warming. If there was any real hazard to it our nanny government would be all over it like brown on poo. Just don't lick your fingers clean afterwards and you'll live forever, I've been casting bullets and fishing sinkers/lures and soldering electronic circuits since 1965 and it's never bothered me. Me. Me. Me. Me.
458winshooter
New member
Lead poison
I use a dust mask, safety glasses,long sleeve shirt and I have a fan blowing across the pot to the outside air.I started casting a little over a year ago and at first I did not think that much about it.After I read some posts here about some of the members being tested for lead in their blood I decided to start taking some precautions.
I use a dust mask, safety glasses,long sleeve shirt and I have a fan blowing across the pot to the outside air.I started casting a little over a year ago and at first I did not think that much about it.After I read some posts here about some of the members being tested for lead in their blood I decided to start taking some precautions.
